Lessons from PROGRESS on biosample collection in digital trials
Authors: Isabel Bruce & Danielle Chiang –
Attrition (dropout) rates pose a barrier to successful and meaningful data collection in clinical trials. One of the promises of decentralized clinical trials (DCTs) is that they can open doors to more diverse participants—without asking them to spend hours driving to clinics or waiting in labs. By minimizing barriers to participation, DCTs have the potential to broaden the scope of clinical trials and enable higher retention rates. But the big question remains: will people actually stick with it and return their samples?
As introduced in previous posts, the Scripps Research Digital Trials Center launched the PRediction Of Glycemic RESponse Study (PROGRESS) to create a predictive model of glucose responses to nutritional intake in people with type 2 diabetes (T2D), diagnosed with prediabetes, or normoglycemic (full disclosure, I was a willing and enthusiastic participant of PROGRESS). A key part of this study involved at-home biological sample collection, as participants were mailed kits that were self-administered for collection of blood, stool, and saliva samples at baseline. These collection kits were chosen after careful consideration and comparison, with an emphasis on participant safety, convenience, and viability–as written about in previous posts.
Benefits of at-home biological sample collection include minimizing geographical barriers to participation and decreasing the associated time commitment for participants. Traditional in-clinic study methods require participants to drive to their local hospital or lab, find (and pay for) parking, wait to be seen, and enter a clinical setting that is likely unfamiliar. At-home biological sample collection provides an opportunity for participants to complete study tasks at their own time, pace, and in a home environment.
In my own experience completing the PROGRESS biosample kits, the entire process took around 20 minutes. This includes time spent “setting up” each kit, taking my samples, and packaging them for data collection and analysis. The blood sample kit, which includes detailed instructions in written, image, and video formats, took me about four minutes to complete, while the stool sample collection similarly required only around 6 minutes. Average time to complete the PROGRESS salivary kit ranges between 2 to 5 minutes (DNAgenotek, 2025). Collecting these samples at home also gave me the flexibility to split up each task, as I completed the blood sample on a Friday morning in pajamas, and the stool sample on a Sunday night before watching a movie. In the time it would have taken me to drive to the nearest hospital clinic, I completed my self-data collection.
Minimizing the time and effort needed to participate in clinical research studies may promote representation for traditionally overlooked and excluded groups. The PROGRESS study’s sample demographics support this claim, demonstrating “how decentralized digital trials allow for continuous capture of contextualized data in real-world settings while ensuring robust representation with a diverse cohort with 48.1% of participants self-identified as UBR [Underrepresented in Biomedical Research]” (Carletti, M., Pandit, J., Gadaleta, M. et al. 2025).
However, looking at initial enrollment isn’t enough to confirm the “success” of the direct-to-participant model. Getting people into the study is a success in and of itself; getting participants to then complete tasks and stay engaged is where the study can truly come to life. Participant data allows us to investigate our research questions and arrive at meaningful and significant conclusions to help move science forward.
To establish the effectiveness of at-home biological sample collection within the PROGRESS study, we conducted a literature review to analyze the PROGRESS data in the context of pre-existing studies. As a secondary interest, we also sought to evaluate whether completion rates varied between the three samples: blood, stool, and saliva.
Our literature review consisted of 15 articles and studies regarding trends in overall attrition and specific metrics for biological sample collection. Studies were chosen on the basis of biological samples collected, sample demographics, and attrition data collected. We intended to create a general picture of clinical trials in today’s landscape, enabling comparison with the PROGRESS study. Reviewed studies included populations with and without type 2 diabetes, as well as both in-person and decentralized designs.
Looking at studies that specifically included patient populations with Type 2 diabetes and employed at-home biological sample collection, our literature review established a range of completion rates between ~ 70-86%. By looking at recruitment, retention, diversity, and satisfaction, our review provided meaningful data concerning decentralized models. Our data suggests the viability of DCTs as alternatives to in-person designs.
To evaluate the PROGRESS completion rates against our literature data, we analyzed at-home biological sample completion for 656 participants from the larger PROGRESS database. Participants were selected on the basis of whether they received a Biokit (containing all three of the sample collection kits). In order to receive these Biokits, participants needed to have first connected their electronic health records in the study app and completed preliminary surveys.
Within the participants who had received Biokits, we assessed completion of each task (blood, stool, and saliva) based on return of sample and feasibility/quality of sample to successfully derive the following results:
- Blood: A1C value
- Stool: Microbiome diversity percentile
- Saliva: ROI Poly risk percentile
Our data analysis showed comparable completion metrics for at-home biological sample collection in PROGRESS versus preexisting clinical studies–looking at both other DCTs and traditional in-clinic studies. Completion rates for at-home biological sample collection in PROGRESS ranged between ~72-77% (Figure 1), whereas our literature review found that clinical studies across the board have an average retention rate of 70%. While the studies we used for comparison with PROGRESS were not exact replicas, we used a range of study models and demographics to create a comprehensive image of trends in clinical studies. Articles reviewed included those with a general focus on self-collection completion rates–like a 2025 paper on the plausibility of “at-home clinics”–as well as more targeted studies on populations with Type 2 diabetes, such as a paper looking at salivary collection in people with T2D across 5 Native American tribes.
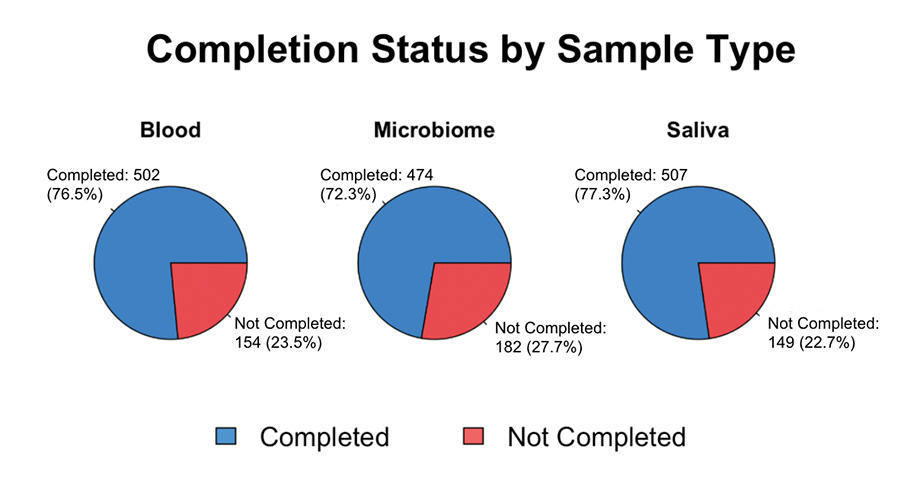
Figure 1. At-home biological sample collection completion rates for participants in the PROGRESS study separated by sample (blood, stool, or saliva). Completion is defined as returning a viable biosample. “Not completed” meant a participant either didn’t return their sample, or the sample couldn’t be used (contents were damaged, sample taken incorrectly, etc.).
To address our second question regarding a potential association between task (sample type) and completion rate, we conducted statistical analysis using a chi-square test (Figure 2). A chi-square test looks for evidence of a statistical association by comparing “observed” data to “expected” data. Expected counts reflect what we would predict given no association between our variables of interest (i.e. there is no correlation between task type and completion). Significant differences between the observed and expected counts indicate a statistical association between the variables of interest–in this case task type (blood, saliva, or stool) and completion.
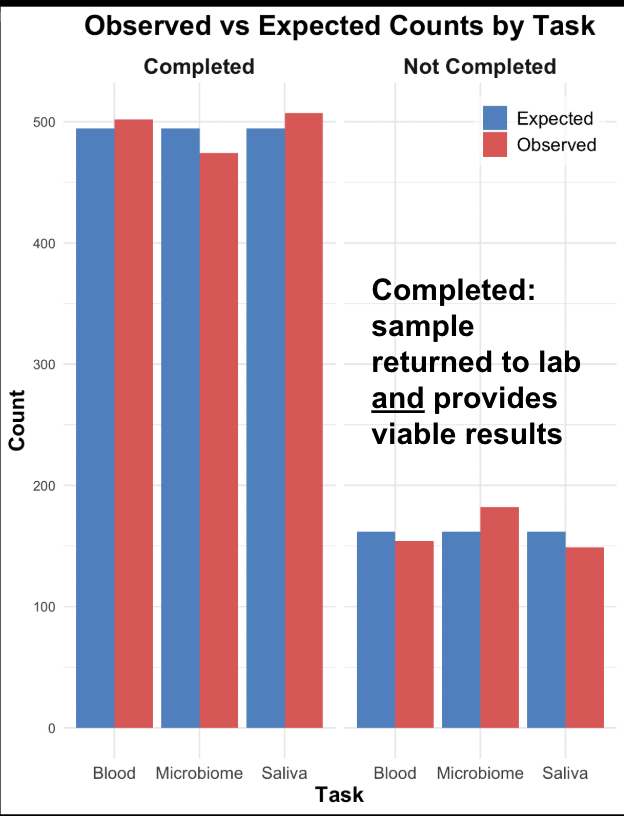
(A) Observed vs. expected counts by task (sample type).
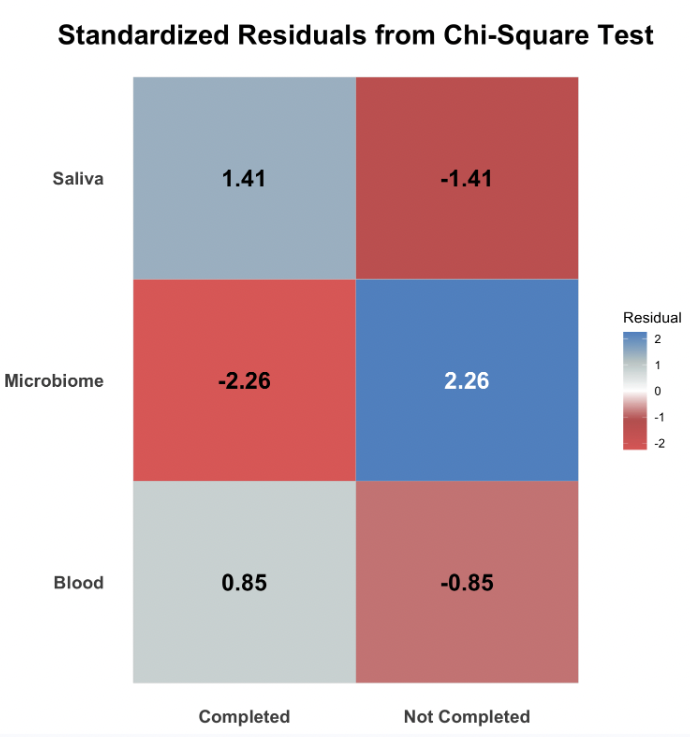
(B) Heat map of standardized residuals from chi-square test.
Figure 2. Results of the chi-square test for an association between task (sample type) and completion rate. (A) The observed vs. expected counts for each task, with the expected counts calculated based on the chi-square test parameters. (B) A heat map of the standardized residuals from the chi-square test, with darker colors indicating a greater absolute value and thus greater deviation from the expected.
Our statistical analysis shows some evidence for an association between sample type (blood, stool, or saliva) and completion rate. More specifically, the stool (or microbiome) sample is associated with lower completion rates. Figure 2A indicates this association, as we see that the actual number of participants who completed the stool sample was lower than the expected count, given no statistical association. Furthermore, Figure 2B shows how much the observed data deviated from the expected–with greater absolute values (as seen for the stool sample) indicating greater evidence for an association between sample type and completion rates.
The potential association between task and completion rate may call for increased incentivization for specific sample types (i.e. the stool sample). Nevertheless, our results reinforce the efficiency and advantages of DCTs–and specifically at-home biological sample collection. The lowest completion rate observed in PROGRESS was 72.3%, and while steps should be taken to continue increasing this percentage, this metric demonstrates successful data collection. As mentioned previously, our literature review estimated the average completion rate across clinical trials at ~70%, meaning PROGRESS saw above-average completion for the at-home biological sample collection tasks.
At-home biological sample collection provides participants with flexibility and autonomy, while offering the same quality data acquired in traditional clinical studies. For researchers, this means reliable, high-quality biosamples without the bottlenecks of in-clinic visits. For participants, it means research that fits into their lives. Together, these findings point to a future where decentralized trials aren’t just feasible–they may outperform traditional methods in retention and representation
What’s next?
As the shift towards direct-to-participant clinical trials continues, this method of self-guided data acquisition can be an invaluable tool in studies seeking representative and sustainable samples. However, to maximize the efficiency and success of at-home biosample collection–we must focus on the participant experience.
One priority is minimizing potential anxiety surrounding proper sample collection and usage. Taking your own blood can be daunting, so it’s crucial to ensure that instructions are digestible and transparent for all biosampling tasks, and that kits are carefully chosen. When selecting the blood sample kit for the PROGRESS study, various considerations were considered–including convenience, safety, packaging, consistency, etc.(Quartuccio, Chiang, & Pawelek, 2022). By listening to participant feedback, testing out new kits and methods of sample collection, and simplifying instructions to ensure accessibility, researchers can continue to increase completion rates and streamline studies in the form of decentralized clinical trials.
References
- Carletti, M., Pandit, J., Gadaleta, M. et al. Multimodal AI correlates of glucose spikes in people with normal glucose regulation, pre-diabetes and type 2 diabetes. Nat Med (2025). https://doi.org/10.1038/s41591-025-03849-7.
- DNA Genotek – Support – Collection Instructions – DNA – RNA – Infectious Disease – Animal Genetics. (2025). Dnagenotek.com. https://www.dnagenotek.com/us/support/collection-instructions/oragene-discover/OGR-500andOGR-600.
- Quartuccio, K., Chiang, D., & Pawelek, J. (2022, February 9). The inner workings of site-less digital clinical trials – Part 2: The at-home blood collection kit – Scripps Research Digital Trials Center. Scripps Research Digital Trials Center. https://digitaltrials.scripps.edu/the-inner-workings-of-site-less-digital-clinical-trials-part-2-the-at-home-blood-collection-kit/.